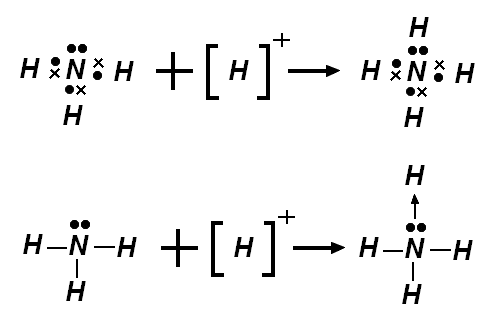[cs_content][cs_section parallax=”false” separator_top_type=”none” separator_top_height=”50px” separator_top_angle_point=”50″ separator_bottom_type=”none” separator_bottom_height=”50px” separator_bottom_angle_point=”50″ style=”margin: 0px;padding: 45px 0px;”][cs_row inner_container=”true” marginless_columns=”false” style=”margin: 0px auto;padding: 0px;”][cs_column fade=”false” fade_animation=”in” fade_animation_offset=”45px” fade_duration=”750″ type=”1/1″ style=”padding: 0px;”][cs_text]Monitoring and Management > 4. The Atmosphere >
Describe the formation of a coordinate covalent bond[/cs_text][/cs_column][/cs_row][/cs_section][cs_section parallax=”false” separator_top_type=”none” separator_top_height=”50px” separator_top_angle_point=”50″ separator_bottom_type=”none” separator_bottom_height=”50px” separator_bottom_angle_point=”50″ style=”margin: 0px;padding: 45px 0px;”][cs_row inner_container=”true” marginless_columns=”false” style=”margin: 0px auto;padding: 0px;”][cs_column fade=”false” fade_animation=”in” fade_animation_offset=”45px” fade_duration=”750″ type=”1/1″ style=”padding: 0px;”][cs_text]
- Covalent bond: A chemical bond formed between two atoms through the sharing of pairs of electrons.
- Single covalent bond: A covalent bond involving the sharing of one pair of electrons.
- Double covalent bond: A covalent bond involving the sharing of two pairs of electrons.
- An oxygen molecule contains a double covalent bond.
- Coordinate covalent bond: A covalent bond in which one of the two atoms bonded supplies all of the shared electrons.
- Coordinate covalent bonds are formed when one atom does not have a complete outer shell, while another atom does have a complete outer shell and has at least one unshared electron pair.
- Once formed, coordinate covalent bonds are indistinguishable from normal covalent bonds.
[/cs_text][/cs_column][/cs_row][/cs_section][cs_section parallax=”false” separator_top_type=”none” separator_top_height=”50px” separator_top_angle_point=”50″ separator_bottom_type=”none” separator_bottom_height=”50px” separator_bottom_angle_point=”50″ style=”margin: 0px;padding: 45px 0px;”][cs_row inner_container=”true” marginless_columns=”false” style=”margin: 0px auto;padding: 0px;”][cs_column fade=”false” fade_animation=”in” fade_animation_offset=”45px” fade_duration=”750″ type=”1/1″ style=”padding: 0px;”][x_video_embed no_container=”false” type=”16:9″][/x_video_embed][/cs_column][/cs_row][/cs_section][/cs_content]